Tell me more about the PD-L1 expression and how cancer uses it to evade our immune response
PD- 1, PD-L1 & the Checkpoint pathway
Programmed cell death protein 1 (PD-1) and its ligand-receptor partner (PD-L1) play a critical role in regulating the immune system's response to cancer. Together they are known as the Checkpoint Pathway.
What are Ligands
Ligands are a type of protein that can be expressed on the surface of any cell
1. Ligands can transmit a message
2. Ligand receptors can receive a message
T-Cells activated by innate immune cells
T cells are our immune system champion warriors or perhaps you can look at them as our nuclear missiles that are activated by our innate immune systems foot soldiers. Essentially a foot soldier cell will alert and prime the T Cells into seek-and-destroy.
What do I mean by "Innate"?
We have two basic parts to our immune system
1. The "Innate" - is our initial / frontline defense
2. The "Adaptive" is our nuclear missiles (T-Cells)
To continue keeping this very simple, if something is not able to be managed by our frontline "innate" immune warriors, then those warriors instead alert the "Adaptive" warriors, and in the context of this article that means alerting the "T-Cells"
Activated T-Cells
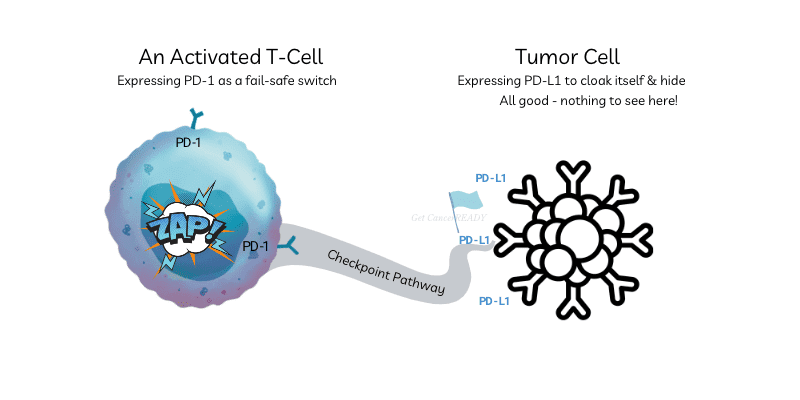
Activated T Cells express the PD-1 protein ligand on their surface. They use PD-1 as a last-minute fail-safe switch to turn off the attack if they encounter a cell expressing PD-L1.
Our immune system is constantly monitoring to eliminate foreign threats, equally, our immune system is also constantly regulating. What can happen (and often does) is the immune system switches into repair mode after the initial T Cell activation. Introducing the PD-L1 checkpoint protein expression is the immune system's way of communicating to the activated T Cells to stand down - everything is now OK.
Immunotherapy
Pharmaceutical companies have developed drugs that can attach to either PD-1 or PD-L1.
By attaching an antibody they are blocking the ability for them to bind. This blocking is called inhibiting and the drug is called an Immune Checkpoint Inhibitor. By preventing the PD-1/PDL1 from binding, the tumor remains uncloaked and unprotected from the activated T Cell attack.
Two Primary strategies
There are more strategies but the following are the primary strategies that we must understand. Other strategies are similar.
The discovery of the checkpoint pathway was made by Jim Allison (USA) and Tosiku Honjo (Japan). They together shared the 2018 Nobel Prize for their discovery.
Pharmaceutical Companies then developed immunotherapy agents (drugs) for clinical trials to discover the most efficient and effective inhibitor treatments.
Strategy 1
Attach an antibody to block PD-1. The most common PD-1 inhibitors are ~
- Pembrolizumab (Keytruda)
- Nivolumab (Opdivo)
Strategy 2
Attach an antibody to block PD-L1. The most common PD-L1 inhibitors are ~
- Durvalumab (Imfinzi)
- Atezolizumab (Tecentriq)
- Avelumab (Bavencio)
The most successful strategy to date is to block PD-1 on the T Cell surface, unfortunately, there has not been the same level of success in blocking PD-L1, although at the time of writing Durvalumab (Imfinzi) is making fast inroads and increasing success outcomes.
These therapies have been approved for the treatment of several types of cancer, including non-small cell lung cancer, melanoma, and renal cell carcinoma.
How do doctors know if this will work on my tumor?
PD-L1 expression is most often evaluated by immunohistochemistry (IHC Test) on tumor tissue, and it's considered a predictive biomarker for response to anti-PD-1/PD-L1 therapy.
PD-L1 expression varies widely among different types of cancer and even among patients with the same type of cancer.
High PD-L1 expression is associated with a better response to therapy, but some patients with low or no PD-L1 expression may still respond to treatment.
It is also important to note that PD-L1 testing is not a definitive tool to predict response to immunotherapy, it's just a way of identifying patients who have a higher likelihood of responding to the treatment. Other factors, such as the tumor mutation burden (TMB) and overall health of the patient, also play a role in determining response to therapy.


Leave A Comment